 
Clearly, the relationship of element properties and atomic weights was a bit more complicated, as Mendeleev soon realized. But Newlands’ pattern did not hold up very well after the first couple of octaves, leading a critic to suggest that he should try arranging the elements in alphabetical order instead. In England, the chemist John Newlands noticed that arranging the known elements in order of increasing atomic weight produced a recurrence of chemical properties every eighth element, a pattern he called the “law of octaves” in an 1865 paper. Other chemists perceived links between atomic weights and chemical properties, but it was not until the 1860s that atomic weights had been well enough understood and measured for deeper insights to emerge. All together, they capture the entire repertoire of known chemistry. Lithium, sodium and potassium were also a triad.Įvery element on this venerated table has its own story. Bromine, for example, had an atomic weight midway between the weights of chlorine and iodine, and all three elements exhibited similar chemical behavior. When contemplating those weights, Döbereiner noted that certain sets of three elements (he called them triads) showed a peculiar relationship. 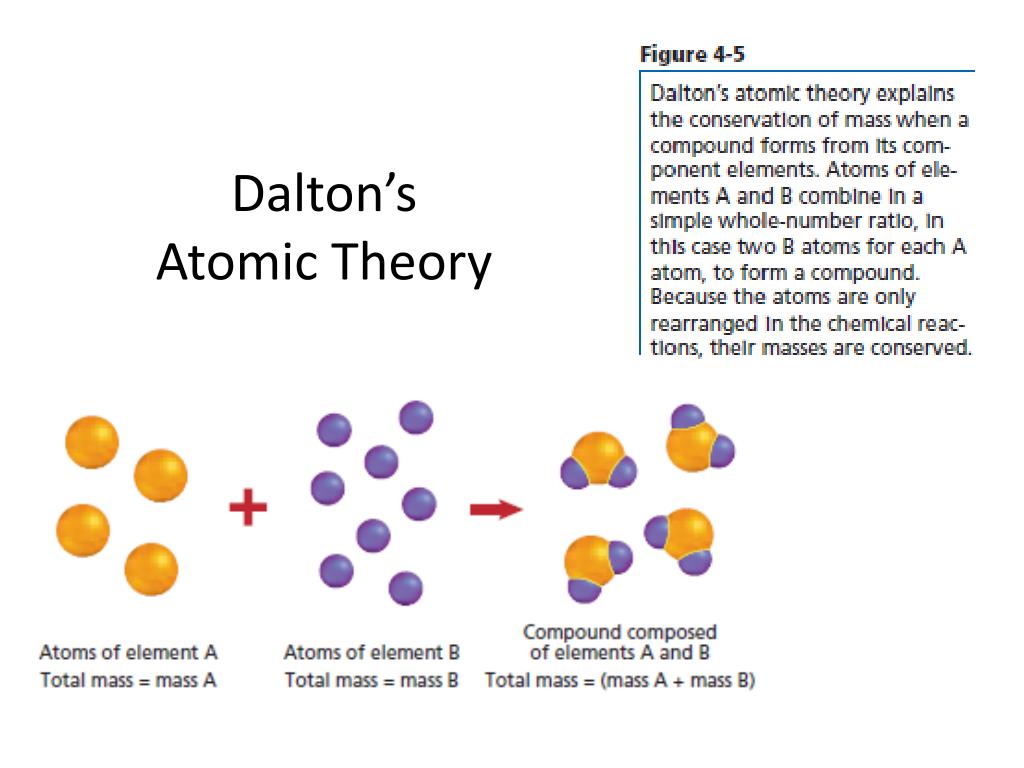
Measuring atomic weights accurately became a prime preoccupation for chemists in the decades that followed. But his theory made the idea of atoms useful, inspiring a revolution in chemistry. When elements combined to make new substances, the amounts that reacted could be calculated with knowledge of those atomic weights.ĭalton was wrong about some of the weights - oxygen is really 16 times the weight of hydrogen, and carbon is 12 times heavier than hydrogen. Oxygen atoms weighed eight times as much as hydrogen atoms carbon atoms were six times as heavy as hydrogen, Dalton believed. Any given element consisted entirely of one kind of atom, he reasoned, distinguished from other kinds by weight. The periodic table symbolizes not merely the constituents of matter, but the logical cogency and principled rationality of all science.Ĭhemical reactions, Dalton proposed, produced new substances when atoms were disconnected or joined. 
His table finished the transformation of chemical science from the medieval magical mysticism of alchemy to the realm of modern scientific rigor. It hinted at the existence of subatomic structure and anticipated the mathematical apparatus underlying the rules governing matter that eventually revealed itself in quantum theory. It validated the then-controversial belief in the reality of atoms. Mendeleev’s table did more than foretell the existence of new elements. “The law of periodicity first enabled us to perceive undiscovered elements at a distance which formerly was inaccessible to chemical vision.” “Before the promulgation of this law the chemical elements were mere fragmentary, incidental facts in Nature,” Mendeleev declared. His law revealed profound familial relationships among the known chemical elements - they exhibited similar properties at regular intervals (or periods) when arranged in order of their atomic weights - and enabled Mendeleev to predict the existence of elements that had not yet been discovered. Mendeleev’s table looked like an ad hoc chart, but he intended the table to express a deep scientific truth he had uncovered: the periodic law. “The periodic table,” wrote the chemist Peter Atkins, “is arguably the most important concept in chemistry.”
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
